| |

307 Chapter 3 Reading Guide 3.1 (14pts)
- Write all answers in your bound journal
- Make it NEAT and in COMPLETE SENTENCES, clearly identify which section you are working (using numbers)
- Box final answers if doing math.
- -4pt if you do not title and date the entry
- -1pt (per infraction) if you do not clearly identify the sections, or number the problems
Answer each question completely (include equations and drawings when asked)
- Who came up with Stoichiometry and in what year was it established?
- What part of Dalton's Atomic Theory gave way to Stoichiometry?
- Give the equation for the formation of water,and circle the products and box the reactants.
- What are the numbers in front of the formulas called? What do they represent?
- Why it the number "1" left out of the equation?
- How many Hydrogen and Oxygen are on each side of the equation?
- How many Mg, O, and H,are represented by the notation 3Mg(OH)2?
- A balanced chemical equation should contain the ____________ possible _______-number ___________.
- If you change the __________ in a formula you change the identity of the substance.
- However when you change the _________ of the formula you change the amount.
- NEVER CHANGE THE _______________ WHEN BALANCING AN EQUATION.
- Write equation [3.2] on page 79
- Illustrate what happens in this equation
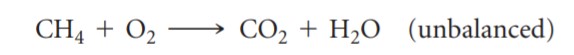
using a circle diagram like this 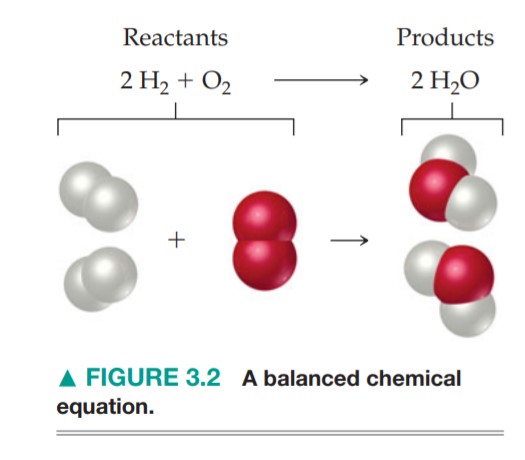
- What do (g), (l), (s), and (aq) represent in a chemcial equation?
|
|

