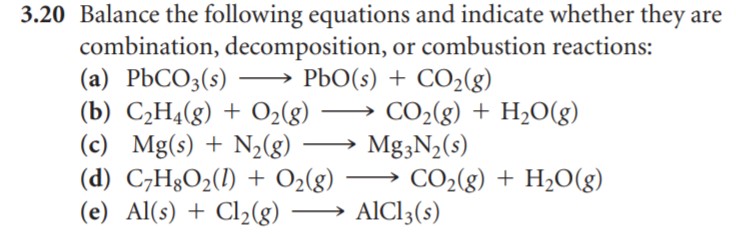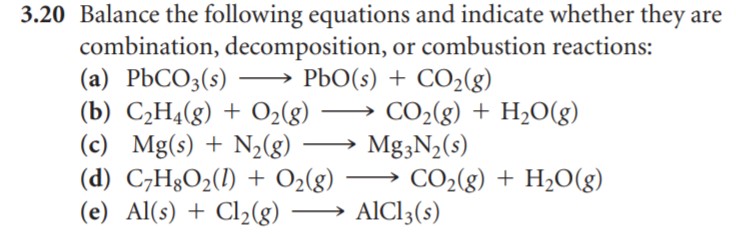| |

308 Chapter 3 Reading Guide 3.2-3 (23pts)
- Write all answers in your bound journal
- Make it NEAT and in COMPLETE SENTENCES, clearly identify which section you are working (using numbers)
- Box final answers if doing math.
- -4pt if you do not title and date the entry
- -1pt (per infraction) if you do not clearly identify the sections, or number the problems
Answer each question completely (include equations and drawings when asked)
- Formula weights 3.1, starts off with the phrase
"chemical formulas and chemical equations both have a quantitative significance..."
what does this phrase mean?
- What is the formula weight of a substance?
- Explain (as they do in the book) the formula weight of H2SO4.
- What is the difference between formula weight and molecular weight?
- What is percent composition of a compound?
- What is the percent composition of Carbon, Hydrogen, and Oxygen in C12H22O11?
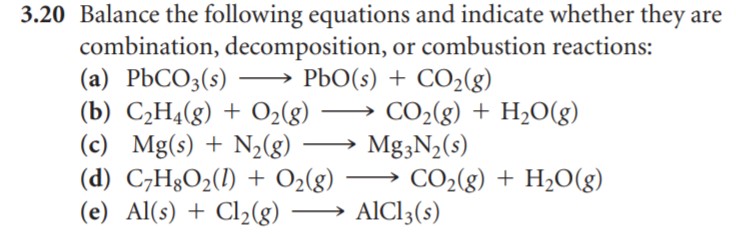


|
|