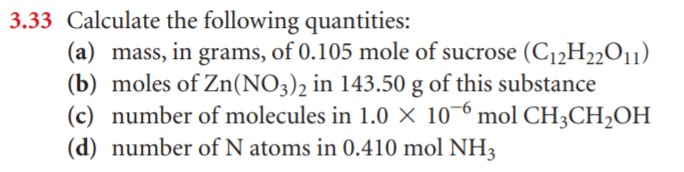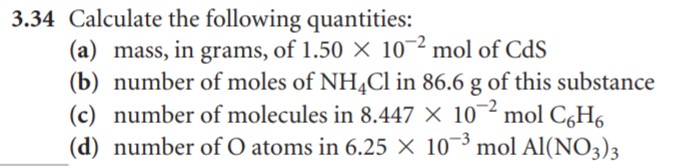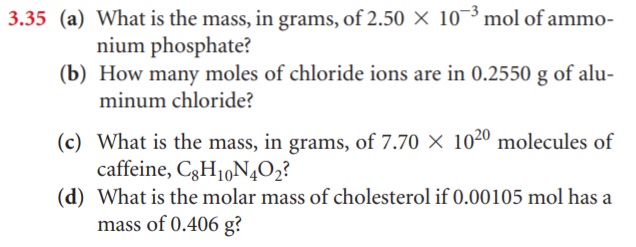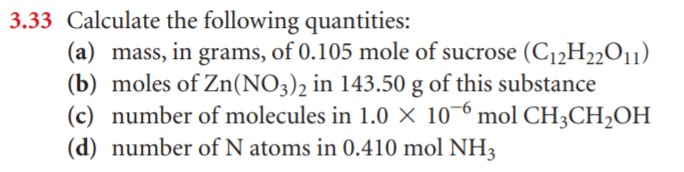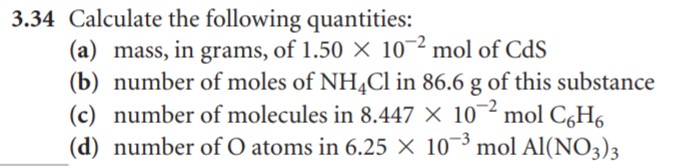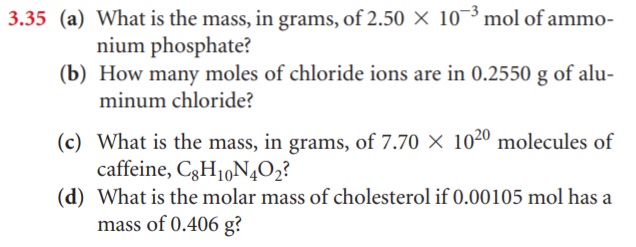| |

310 Chapter 3 Reading Guide 3.4 (29 pts)
- Write all answers in your bound journal
- Make it NEAT and in COMPLETE SENTENCES, clearly identify which section you are working (using numbers)
- Box final answers if doing math.
- -4pt if you do not title and date the entry
- -1pt (per infraction) if you do not clearly identify the sections, or number the problems
Answer each question completely (include equations and drawings when asked)
- What has 2x1023 water molecules?
- What is a mole and what is it based on?
- Why was the "mole unit" invented by chemist?
- What is the 6.0221421x1023 represent, and WHO came up with this number?
- What visual could be used to represent 1 mole of marbles?
- What visual could be used to represent 1 mole of pennies?
- Without using a calculator, arrange 12g 12C, 1 mol of C2H2 and 9x1023 molecules of CO2 in increasing number of carbon atoms.
- Calculate the number of H atoms in 0.350 mol of C6H12O6.
- What is the relationship between molar mass of an atom and AMU (use H2O to explain)?
- Calculate the number of moles of glucose (C6H12O6) in 5.30g of C6H12O6.
- Calculate the number of grams in 0.350 mol of C6H12O6.
- Calculate the number of glucose molecules in 5.23g of C6H12O6.
- How many oxygen atoms are present in this sample?
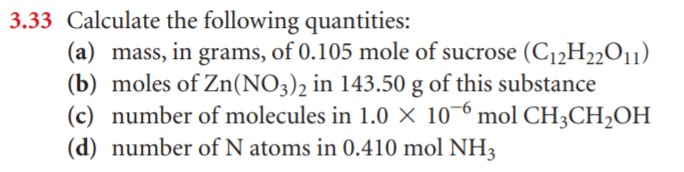
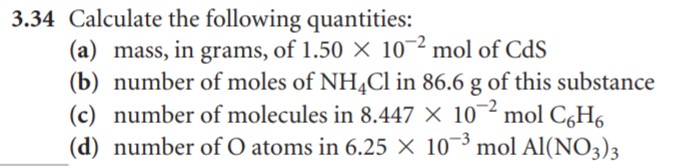
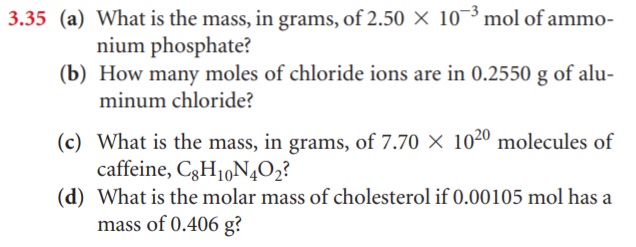

|
|