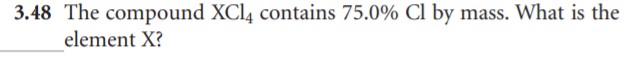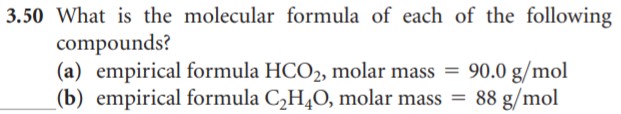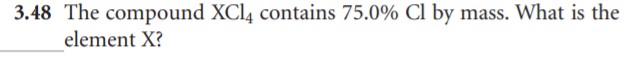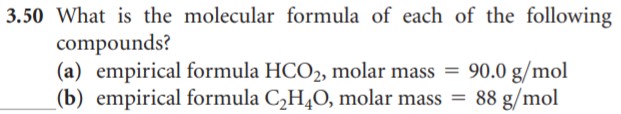| |

311 Chapter 3 Reading Guide Section 5 (25pts)
- Write all answers in your bound journal
- Make it NEAT and in COMPLETE SENTENCES, clearly identify which section you are working (using numbers)
- Box final answers if doing math.
- -4pt if you do not title and date the entry
- -1pt (per infraction) if you do not clearly identify the sections, or number the problems
Answer each question completely (include equations and drawings when asked)
- Determine how many moles of Mercury and Chlorine are present in a 100.0g sample
that is 73.9% Mercury and 26.1% Chlorine by mass.
- Using the information above, determine the mole ratio of Mercury Chloride.
- What must be the chemical formula for Mercury Chloride based on your data?
- If ascorbic acid is 40.92% C, 4.58% H and 54.90% O by mass, what is the empirical formula?
- Explain how to detemine the molecular weight of acorbic acid.
- What is combustion analysis?
- Isopropyl alcohol is composed of C, H, O. Combustion of a 0.255 gram sample produces
0.561g of carbon dioxide and 0.306g of water. Determine the empirical formula.








|