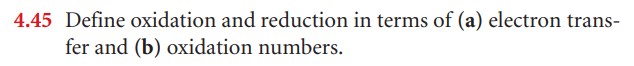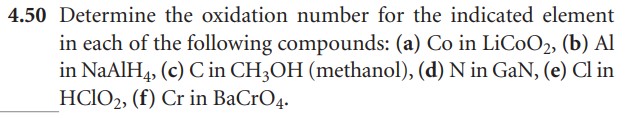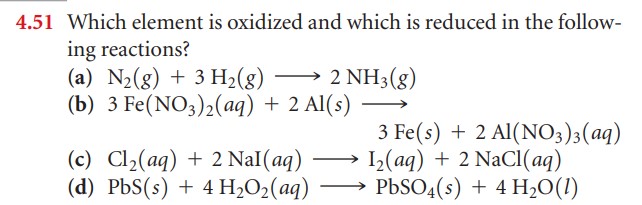| |

416 OXIDATION AND REDUCTION (book SECTION 4.4)
- Write all examples and solutions in your bound journal
- Make it NEAT and in COMPLETE remember to keep your units with all numbers
- -4pt if you do not title and date the entry
- -1pt (per infraction) if you do not clearly identify the sections, or number the problems
What is oxidation and reduction?
An oxidation-reduction (redox) reaction is a type of chemical reaction that involves a transfer of electrons between two species. An oxidation-reduction reaction is any chemical reaction in which the oxidation number of a molecule, atom, or ion changes by gaining or losing an electron.
Simply put...if you gain an electron, you are reduced!
(becoming more negative is reduced)
If you lose an electron, you are oxidized!
Redox reactions are common and vital to some of the basic functions of life, including photosynthesis, respiration, combustion, and corrosion or rusting.
Rules for Assigning Oxidation States
The oxidation state (OS) of an element corresponds to the number of electrons, e-, that an atom loses, gains, or appears to use when joining with other atoms in compounds. In determining the OS of an atom, there are seven guidelines to follow:
- The OS of an individual atom is 0.
- The total OS of all atoms in: a neutral species is 0 and in an ion is equal to the ion charge.
- Group 1 metals have an OS of +1 and Group 2 an OS of +2
- The OS of fluorine is -1 in compounds
- Hydrogen generally has an OS of +1 in compounds, except with a metal it is -1 (e.g. NiH2)
- Oxygen generally has an OS of -2 in compounds
- In binary metal compounds, Group 17 elements have an OS of -1, Group 16 of -2, and Group 15 of -3.
(Note: The sum of the OSs is equal to zero for neutral compounds and equal to the charge for polyatomic ion species.)
Example 1: Assigning OSs
Determine the OSs of the elements in the following reactions:
- Fe(s)+O2(g) → Fe2O3(g)
- Fe2+
- Ag(s)+H2S → Ag2S(g)+H2(g)
SOLUTIONS (highlight to see the answers)
- Fe and O2 are free elements; therefore, they each have an OS of 0 according to Rule #1. The product has a total OS equal to 0, and following Rule #6, O has an OS of -2, which means Fe has an OS of +3.
- The OS of Fe corresponds to its charge; therefore, the OS is +2.
- Ag has an OS of 0, H has an OS of +1 according to Rule #5, S has an OS of -2 according to Rule #7, and hence Ag in Ag2S has an OS of +1.
Example 2: Assigning OSs
Determine the OS of phosphorous element in each of the following:
- Na3PO3
- H2PO4-
SOLUTIONS (highlight to see the answers)
- The oxidation numbers of Na and O are +1 and -2. Because sodium phosphite is neutral, the sum of the oxidation numbers must be zero. Letting x be the oxidation number of phosphorus, 0= 3(+1) + x + 3(-2). x=oxidation number of P= +3.
- Hydrogen and oxygen have oxidation numbers of +1 and -2. The ion has a charge of -1, so the sum of the oxidation numbers must be -1. Letting y be the oxidation number of phosphorus, -1= y + 2(+1) +4(-2), y= oxidation number of P= +5.
Example 3: Identifying Reduced and Oxidized Elements
Determine which element is oxidized and which element is reduced in the following reactions (be sure to include the OS of each):
- Zn + 2H+ → Zn+2 + H2
- 2Al + 3Cu+2→ 2Al+3 +3Cu
- CO3-2 + 2H+→ CO2 + H2O
- 2H2 + O2 → 2H2O
- 2H2O → 2H2 + O2
- Cl2 + NaBr → NaCl + Br2
SOLUTIONS (highlight to see the answers)
- Zn is oxidized (Oxidation number: 0 →+2); H+ is reduced (Oxidation number: +1 → 0)
- Al is oxidized (Oxidation number: 0 → +3); Cu+2 is reduced (+2 → 0)
- This is not a redox reaction because each element has the same oxidation number in both reactants and products: O= -2, H= +1, C= +4.
- The OS of H changes from +1 to 0, and the OS of Zn changes from 0 to +2. Hence, Zn is oxidized and acts as the reducing agent, and H is reduced and acts as the oxidizing agent
- Calculation: 0 + 0 ? (2)(+1) + (-2) = 0
Explanation: In this equation both H2 and O2 are free elements; following Rule #1, their OSs are 0. The product is H2O, which has a total OS of 0. According to Rule #6, the OS of oxygen is usually -2. Therefore, the OS of H in H2O must be +1.
- Calculation: (2)(+1) + (-2) = 0 → 0 + 0
Explanation: In this reaction, water is "decomposed" into hydrogen and oxygen. As in the previous example the H2O has a total OS of 0; thus, according to Rule #6 the OS of oxygen is usually -2, so the OS of hydrogen in H2O must be +1.
- Calculation: (0) + ((+1) + (-1) = 0) -> ((+1) + (-1) = 0) + 0
Explanation: In this equation, Br is replaced with Cl, and the Cl atoms in Cl2 are reduced, while the Br ion in NaBr is oxidized.
PROBLEMS
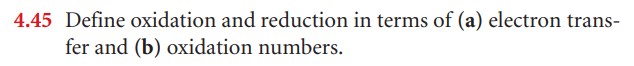
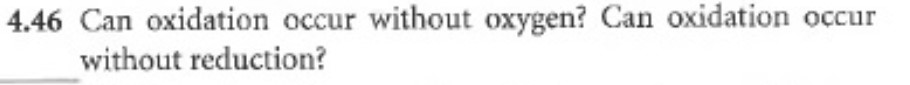



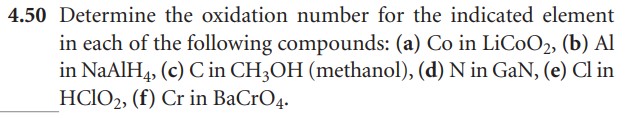
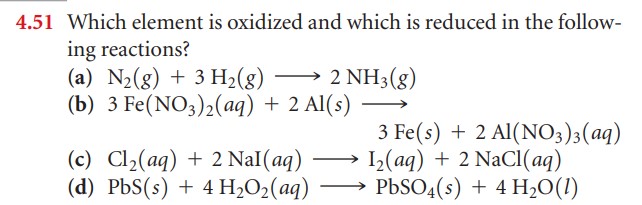





|