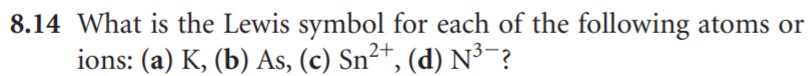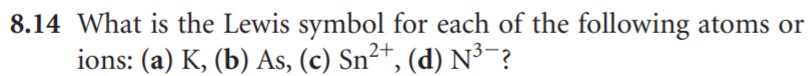| |

801 SECTIONS 1 & 2
- Write all answers in your bound journal
- Make it NEAT and in COMPLETE SENTENCES, clearly identify which section you are working (using numbers)
- Box final answers if doing math.
- -4pt if you do not title and date the entry
- -1pt (per infraction) if you do not clearly identify the sections, or number the problems
Section 1 (11pts)
- What are the three types of bonds
- What is the Lewis symbol for an element?
- What is the octet rule?
- Write the Lewis structure of Oxygen, Phosphorous and Sulfur. Who is similar and why?

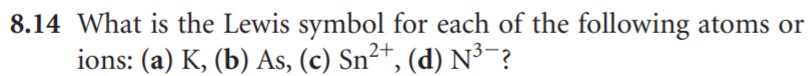
Section 2 (14pts)
- What kind of element combinations form ionic bonds
- Diagram Sodium and chlorine forming an inic bound using Lewis Structures.
- What are characteristics of ionic bonds?
- Is the formation of sodium chloride is exo or endo, what is the value and why?
- Explain mathmatically how sodium chloride crystal formation is exothermic?
- What is lattice energy, why are the values positive?




|