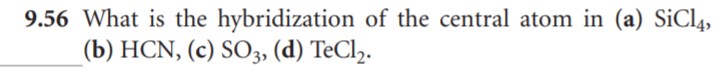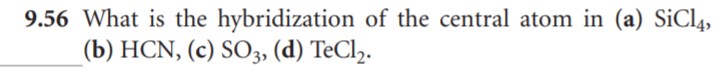| |

903 Section 4 & 5 (15 pts)
- Write all answers in your bound journal
- Make it NEAT and in COMPLETE SENTENCES, clearly identify which section you are working (using numbers)
- Box final answers if doing math.
- -4pt if you do not title and date the entry
- -1pt (per infraction) if you do not clearly identify the sections, or number the problems
READING QUESTIONS
- What is valence bond theory?
- Explain the bonding of HCL using the overlap of atomic orbitals with and illustration.
- Explain what orbital in Chlorine and which orbital in Hydrogen participate in the bond in HCL.
- What are hybrid orbitals?
- Using the molecule BeF2, explain sp hybridization using electron box diagrams.
- Using the molecule BF3, explain sp2 hybridization using electron box diagrams.
- Using the molecule CH4, explain sp3 hybridization using electron box diagrams.
.

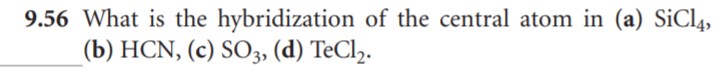

|