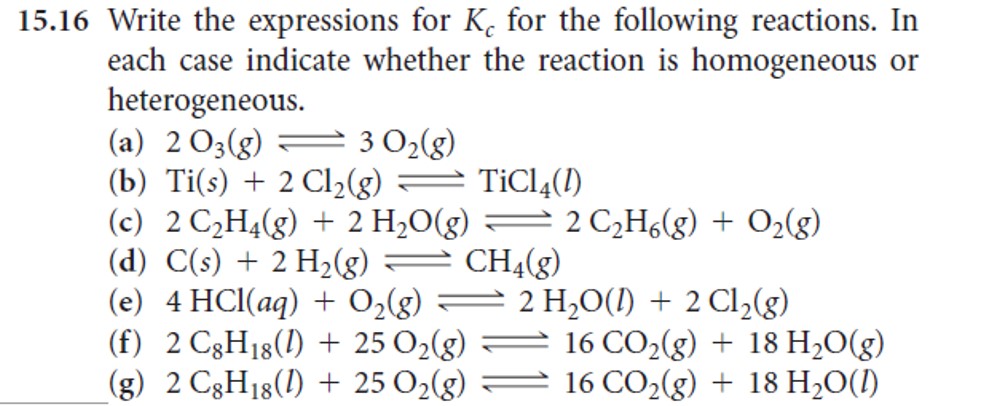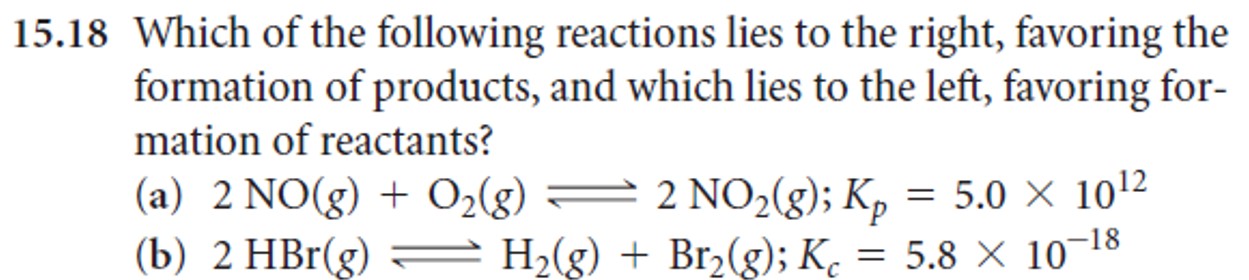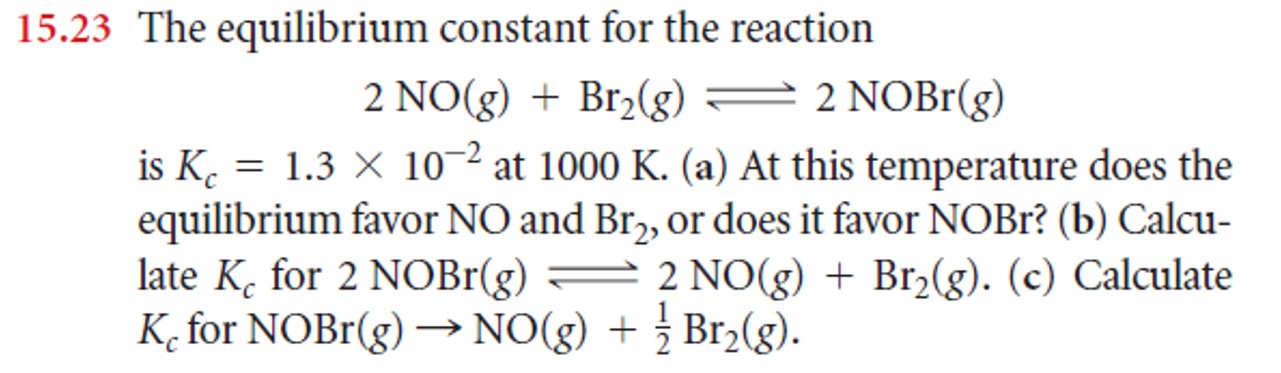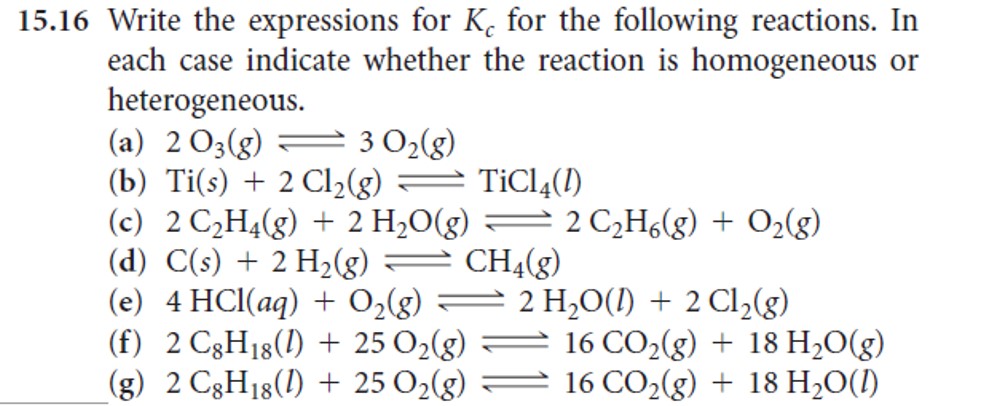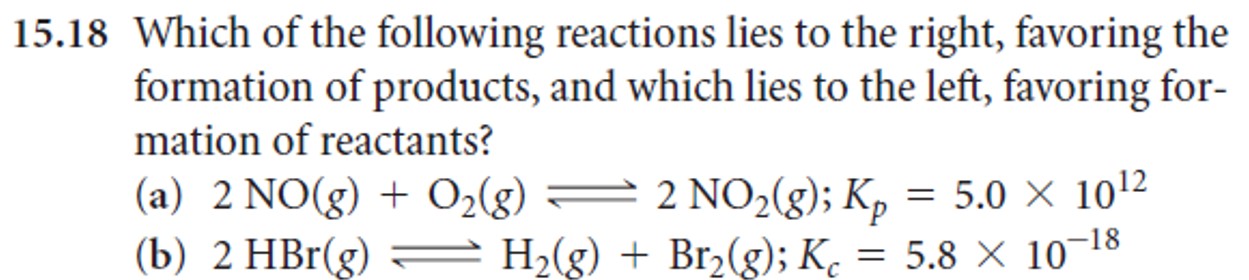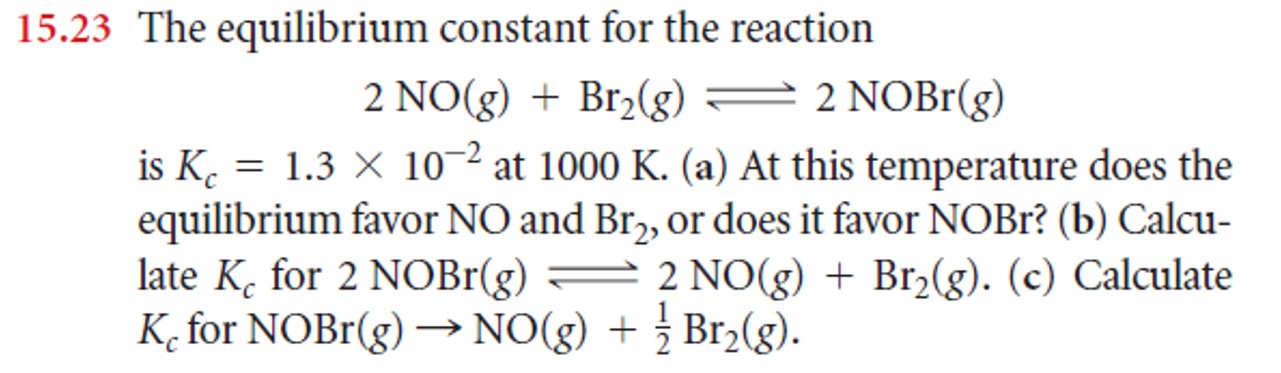| |

1501 Chapter 15: Chemical Equilibrium Outline and Problems:
- Write all answers in your bound journal
- Make it NEAT and in COMPLETE SENTENCES, clearly identify which section you are working (using numbers)
- Box final answers if doing math.
- -4pt if you do not title and date the entry
- -1pt (per infraction) if you do not clearly identify the sections, or number the problems
Section 1 & 2 (28 pts)
- What is chemical equilibrium?
- Explain this concept in regard to N2O4 and NO2 (figure 15.1).
- What are the three concepts (bulleted) on page 613 in regard to equilibrium?
- What is the Haber Process?
- What is the law of mass action? Who came up with it?
- What is the equation for the equilbrium-constant expression?
- How is the Kc defined for the Haber process?
- Complete the sample problems on page 616, be sure to comprehend, how the equilbrium-constant expression are created.
- Using the data from Table 15.1, determine the equilbrium-constant for N2O4 and NO2.
- What does Kp signify?
- What is the ideal gas law (now you know why we backed up to do Chapter 10 befoe this section)?
- What is the equation to derive PA? How is this useful?
- What is the general expression relating Kp and Kc?
- When does Kp = Kc?
- Problems:

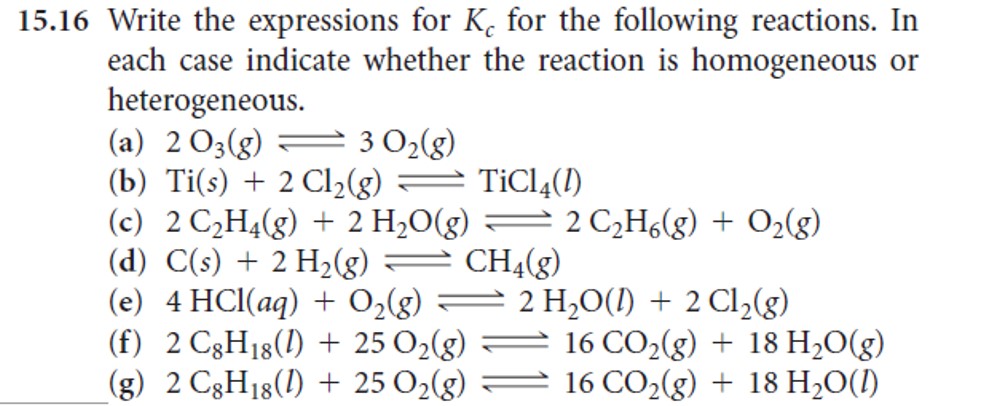
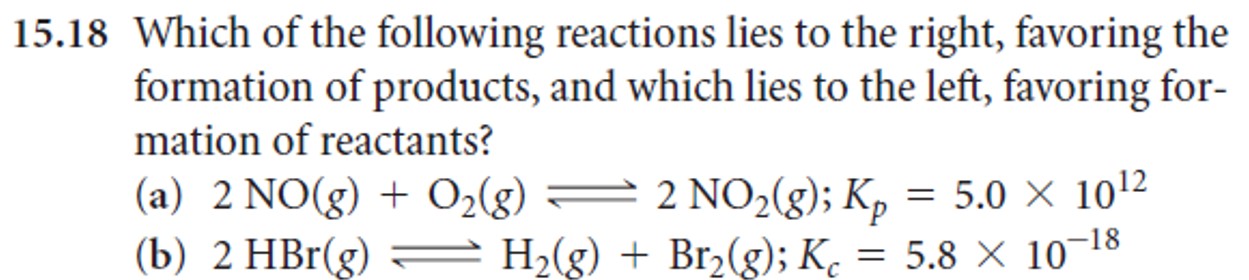

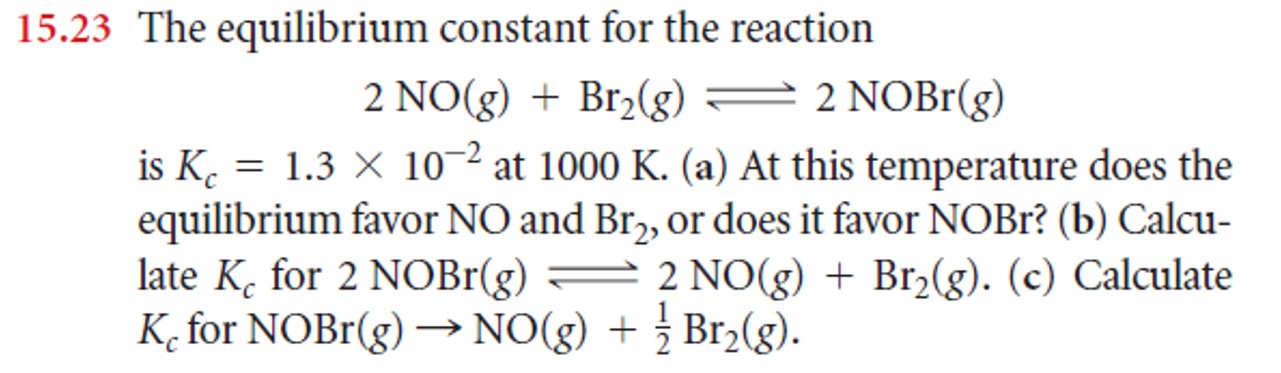
|