| |

1502 Chapter 15: Chemical Equilibrium Outline and Problems:
- Write all answers in your bound journal
- Make it NEAT and in COMPLETE SENTENCES, clearly identify which section you are working (using numbers)
- Box final answers if doing math.
- -4pt if you do not title and date the entry
- -1pt (per infraction) if you do not clearly identify the sections, or number the problems
Section 3 & 5 (20pts)
- Why is there no units with equilibrium constants?
- What is the magnitude of the equilibrium constants?
- What does K>>1 and K<<1 mean?
- What are the Kc for the forward and reverse reactions of N2O4 ⇄ 2 NO2?
- What are the Kc for the forward and reverse reactions of 2 N2O4 ⇄ 4 NO2?
- Explain how to derive the Kc for 2 NOBr(g) + Cl2(g) ⇄ 2 NO(g) + 2BrCl(g).
- What is Homogenous and Heterogenous Equilibria?
- Give and example of Heterogenous Equilibria?
- What isn't included in Equilibria equations?
- What is the Kc and Kp CaCO3(s) ⇄ CaO2(s) + CO2(g)?
- What is the Kc and Kp H2O(l) + CO32-(aq) ⇄ OH-(aq) + HCO3-(aq)?
- Write the five steps for calculating the equilbirium constant if all the final concentrations are known.
- Write out the example problem:
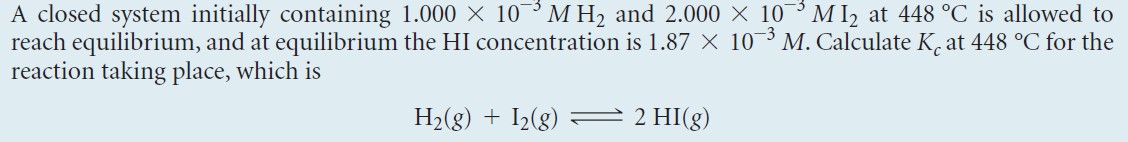


|

