| |

1601 ACID BASE EQUILIBRIA PART 1
(23pts Reading Guide)
(15pts for problem set 1: below)
- Write all information in your bound journal
- Make it NEAT and in COMPLETE SENTENCES remember to keep your units with all numbers
- You can use table format
- Box final answers if doing math.
- -4pt if you do not title and date the entry
- -1pt (per infraction) if you do not clearly identify the sections, or number the problems
- What causes sour taste?
- What is the chemical formula for citric acid, malic acid, oxalic acid, where are these acids found in nature?
16.1
- What was the earliest definitions of acids and bases?
- What was Svante Arrhenius's definition?
- Show mathematically the 37% HCl solution is 12M.
16.2
- What is the Bronsted-Lowry definition of acids and bases, how is it different than Arrhenius?
- Diagram what a H+ ion would do in water. How do chemist note it?
- Explain using the Bronsted-Lowry method HCl in Water.
- Explain using the Bronsted-Lowry method Ammonia in Water.
- What does Amphiprotic mean?
- Explain conjugate acid and conjugate bases using nitrous acid and water.
- Explain conjugate acid and conjugate bases using ammonia and water.
- Explain the relative strengths of acids and bases (3 points).
16.3
- What does auto ionization of water mean?
- Explain how the io-product constant of water s 1.0e-14.
- What is meant if the solution is neutral in terms of concentration?
- What is the concentration of H+ ions if the concentration of OH- is 0.010M?
16.4
- Explain the pH Scale?
- If the concentration of H+ is 1e-3 what is the pH?
- What is the pH of normal human blood?
- What is pOH?
- If the pOH is 10.24 what is the concentration of H+
- What are the 7 color indicator dyes used for determining pH?
(15pts) Problem set1:
16.18, 16.30, 16.36, 16.38
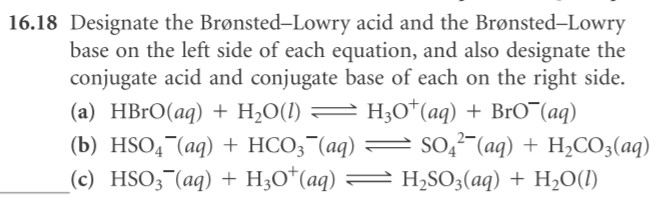



|

