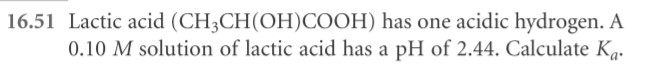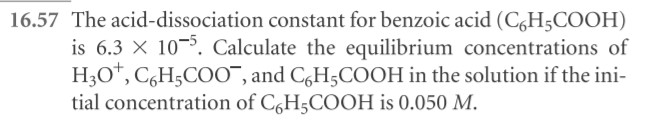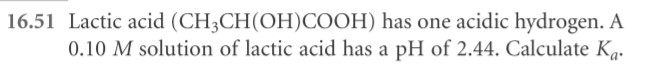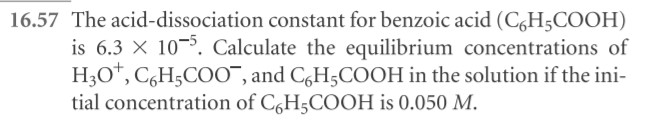| |

1602 ACID BASE EQUILIBRIA PART 2
16.5 (17pts)
- What are strong acids and bases in aqueous solutions?
- What are the seven most common strong monoprotic acids and the one diprotic acid?
- Write the eqautuon for nitric acid and water.
- What is used interchangeably to represent the hydrated proton in water?
- How do you calculate the pH of a strong monoprotic acid?
- What are the strong bases?
- Write out sample exercise 16.9.
- Explain what happens when a solution formed by dissolving 0.010 mol of Na2O(s) in enough water to form
1.0 L solution.
- problems 9pts



16.6 (20pts)
- What is the general equation for a weak acid?
- Why is H2O not in the eqaution?
- What are two different ways to write the equilibrium-constant expression?
- What does Ka signify?
- The magnitude of Ka indicates the __________________________________________: The larger the value of Ka, the __________________________. For most weak acids, Ka values range from ____________ to ____________.
- Follow sample exercise 16.10, and complete this problem. A student prepared a 0.10 M solution of formic acid and found its pH at to
be 2.38. Calculate Ka for formic acid at this temperature.
- What is percent ionization?
- What is percent ionization of a 0.035 M solution of HNO2 which contains 3.7x10-3 M of H+?
- On page 670, write out the steps for calculating Ka from pH.
- In the example, write out how the % ionization was calculated.
- As a general rule, if x is more than about ______% of the initial concentration value, it is better to use the quadratic formula. You should always check the validity of any simplifying assumptions after you have finished solving a problem.
- Higher the pH the ______ acidic.
- The pH of 0.30M Acetic acid is _________. The pH of 0.30M HCL is ___________.
- Doubling the concentration fo a weak acid ____________ double the concentration of H+ ions.
- Calculate the percent ionization of a) 0.10M HF and b) 0.010M HF (hint page 673).
- What is a polyprotic acid?
- Using H2SO3 as an example, show the two equilibrium equations and their Ka values.
- Why would Ka2 always be less than Ka1?
- It is always easier to remove the _______ proton of a polyprotic acid than the _______ one.
- Write out what occurs for dissociation with sulfuric acid.
16.6 PROBLEMS (18pts)

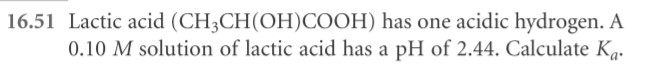


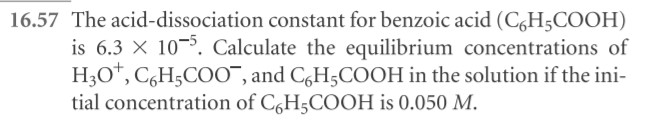

|