| |

1603 ACID BASE EQUILIBRIA PART 3
16.7 (9pts)
- Define a weak base.
- Write the equilibrium constant expression for ammonia.
- Define Kb
- Calculate the concentration of OH- in a 0.15M solution of ammonia.
- What are the two general classes of weak bases? Give an example of each.
- What are the number of moles of NaClO needed to create a 2.0L solution with a pH of 10.50 (show your work)?


16.8 (15pts)
- Using ammonia and water, explain the relationship between Ka & Kb and how it relates to water
- What is pKa & pKb?
- If the Ka for a HF is 6.8x10-4 what is the Kb for the flouride ion?
- If the Ka for a Chlorous acid is 1x10-2 what is the Kb for the Chlorite ion?


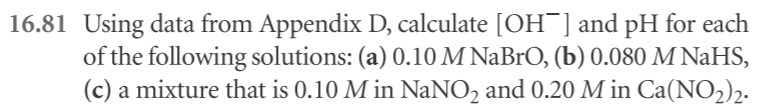

16.9: write out the sentences with the proper choice of word)
- In the acid HA what is A- considerd (conjugate acid or conjugate base?)
- if the anion (A-) reacts with water it will produce what polyatomic ion?
- In the 7 strong acids the Anion (A-) produces a (negligable or excessive) tendency to produce OH- ions?
- If HA is not one of the strong acids then the A- will definitely form a (weak or strong) base in solution?
- This will cause the pH of the water to (increase or decrease)?
- For example if CH3COO-(aq) is added to water then end product would be -->
- NH4+(aq) will act like a (acid or base) in solution and will (dontate or accept) a proton to form (H30+ or OH-).
- How does a hydrated Fe3+ ion act as an acid? Does this make sense?
- Draw a diagram showing a hydrated Fe3+ ion producing (H30+ ions in solution.
- What are the possible combinations of a salt forming an acidc, basic, or ndeutral solution when dissolved in water.
- Anion (N) and Cation (N)= WHAT HAPPENS?
(who qualifies and give an example)
- Anion (makes OH-) and Cation (N)= WHAT HAPPENS?
(who qualifies and give an example)
- Anion (N) and Cation (H30+)= WHAT HAPPENS?
(who qualifies and give an example)
- Anion (makes OH-) and Cation (H30+)= WHAT HAPPENS?
(who qualifies and give an example)




|